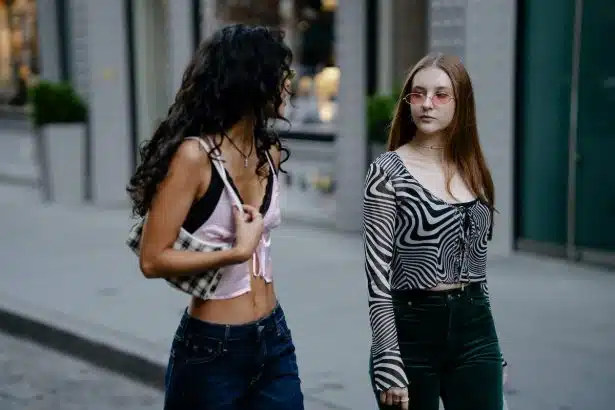Décryptage des tenues de stars : explorez les looks incontournables sur le tapis rouge lors des grands événements prestigieux
Plongeons dans l'univers glamour du tapis rouge où se dévoilent les tenues les plus audacieuses et éblouissantes de nos célébrités…
Beauté
Buzz Cut : secrets de la coupe courte masculine et conseils d’entretien
La coupe buzz cut, un classique intemporel du grooming masculin, revient sur le devant de la scène avec une popularité…
Tout sur le carré de musc : bienfaits, utilisation et achat
Le carré de musc, une substance aromatique prisée depuis l'Antiquité, continue de captiver par son parfum envoûtant et ses vertus…
Guide ultime pour marier les couleurs dans vos tenues : Création d’associations harmonieuses et originales
L'art de l'habillement repose sur une multitude de facteurs, dont l'un des plus importants est sans conteste la maîtrise de…
Mode
Shopping
Chaussons en cuir Bibalou pour bébé : découvrez confort et qualité
Les premiers pas d'un enfant sont un moment précieux et les parents…
Trouvez les meilleures adresses shopping pour des pièces tendances et de qualité en ligne ou en boutique
Que vous soyez un amateur de mode ou simplement à la recherche…
Les incontournables de la mode de la saison : découvrez les tendances à suivre !
À chaque changement de saison, les tendances mode évoluent, nous offrant une…
Les accessoires essentiels pour sublimer votre tenue
Dans le monde de la mode, les vêtements ne sont que la…
Les tendances mode incontournables de la saison
Alors que les saisons changent, la mode évolue également. À chaque nouvelle…
Donnez une nouvelle vie à votre garde-robe grâce à ces astuces ingénieuses
Dans l'univers effervescent de la mode, les tendances vont et viennent, laissant…
Création d’un style unique : astuces pour se démarquer de la masse
Dans un monde empreint de conformisme, l'individualité est devenue un luxe. Chacun…
Guide ultime pour acheter des vêtements d’occasion et vintage de qualité
Plongez dans l'univers fascinant des vêtements d'occasion et vintage de qualité avec…
Les essentiels indispensables pour un dressing féminin parfait
Dans le royaume de la mode féminine, l'harmonie d'un dressing parfait évoque…
Les tendances incontournables des chaussures pour la saison actuelle
Dans le domaine de la mode, le changement est la seule constante.…
Les indispensables pour un style chic et décontracté
Dans une ère où l'apparence occupe une place pivotale, le style chic…
Adopter le style streetwear : conseils pour une allure chic et tendance
Le style streetwear, jadis réservé aux jeunes des zones urbaines, a gagné…
Découvrez les marques engagées pour une consommation responsable
Dans un monde où la surconsommation et le gaspillage sont devenus monnaie…
Les clés pour une garde-robe capsule idéale : conseils et astuces essentiels
Dans le monde de la mode, une tendance durable fait son chemin…
Les accessoires indispensables pour sublimer votre tenue et afficher un style tendance
Dans un monde où l'apparence joue un rôle crucial, où la première…
Découvrez les incontournables de la mode cette saison
Alors que l'automne s'installe doucement, la mode évolue et se transforme, révélant…